Medical Devices: Page 7
-
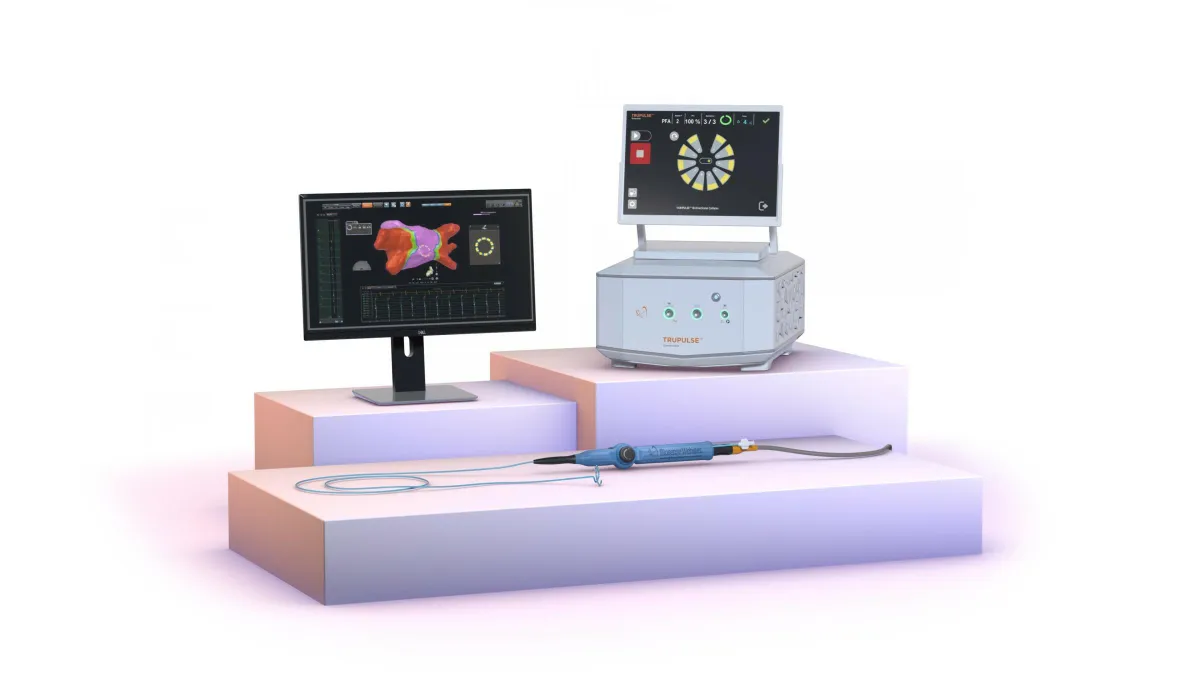
J&J pauses Varipulse PFA cases in US
The temporary halt to investigate reported neurovascular events comes two months after Johnson & Johnson won FDA approval for the pulsed field ablation system.
By Susan Kelly • Jan. 8, 2025 -
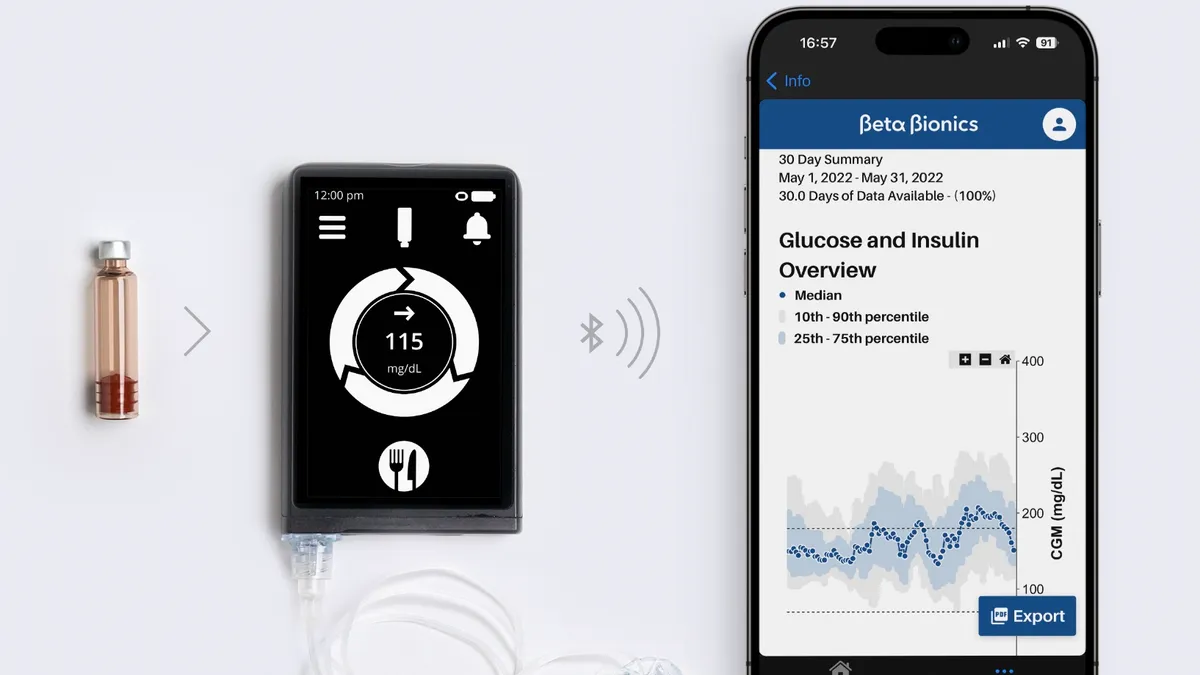
Beta Bionics, maker of ‘bionic pancreas,’ files for IPO
Beta Bionics did not disclose the number of shares it will offer or a price range.
By Elise Reuter • Jan. 8, 2025 -
 Explore the Trendline➔
Explore the Trendline➔
 Sitthiphong via Getty Images
Sitthiphong via Getty Images Trendline
TrendlineMedical device industry continues to turn to AI
While the industry continues to embrace artificial intelligence, there are still questions about how the new technologies need to be regulated and if they are effective.
By MedTech Dive staff -
Boston Scientific to buy remaining Bolt Medical stake for up to $664M
The deal for Bolt, developer of an intravascular lithotripsy system to treat coronary and peripheral artery disease, continues Boston Scientific’s brisk run of acquisitions.
By Susan Kelly • Jan. 8, 2025 -
FDA finalizes guidance on device shortage reporting rules
The final version adds information on the devices affected by the guidance and the impact of revised legislation on voluntary notifications.
By Nick Paul Taylor • Jan. 8, 2025 -
FDA aims to stem AI device bias, boost transparency in draft guidance
The document follows the agency’s first digital health advisory meeting and a final guidance on postmarket updates to AI devices.
By Elise Reuter • Jan. 7, 2025 -
Outset Medical to pay down debt after raising nearly $300M
Stifel analyst Rick Wise said the financing, a combination of private stock placement and new loans, should help the dialysis device maker return to revenue growth.
By Susan Kelly • Jan. 7, 2025 -
FDA posts early alert about safety risk of Medline’s fluid delivery sets
The agency issued the alert as part of a pilot to improve the timeliness of communications about actions it believes are likely to be high-risk recalls.
By Nick Paul Taylor • Jan. 6, 2025 -
Sponsored by Altium365
Shift left today: Electronics procurement visibility for reliable device development
Periods of uncertainty test the resolve of even the most well-oiled medical device supply chains.
Jan. 6, 2025 -
Olympus begins CEO search after Kaufmann resignation
Stefan Kaufmann, former CEO of Tokyo-based Olympus, stepped down in October after the company investigated an allegation that the executive purchased illegal drugs.
By Susan Kelly • Jan. 3, 2025 -
FDA closes 2024 with string of early alerts on device safety risks
Olympus advised providers to stop using an endoscope accessory due to an infection risk linked to 120 injuries and one death.
By Susan Kelly • Jan. 3, 2025 -
FDA-commissioned review finds no safety concerns linked to tampons
Officials commissioned the independent review in September in response to a study that found tampons may expose users to metals.
By Nick Paul Taylor • Jan. 3, 2025 -
Abbott, Dexcom settle glucose monitor patent lawsuits
The rival companies agreed not to sue each other for the next 10 years.
By Elise Reuter • Jan. 2, 2025 -
Q&A
Few medical devices are designed for children. An FDA-Children’s National collaboration aims to change that.
Kolaleh Eskandanian, chief innovation officer at Children’s National Hospital, said the partnership is meant to address the many challenges with developing devices for children and infants.
By Elise Reuter • Jan. 2, 2025 -
Top 10 medtech deals of 2024
Multibillion-dollar buyouts returned last year, led by Johnson & Johnson’s $13.1 billion acquisition of Shockwave Medical.
By Nick Paul Taylor • Jan. 2, 2025 -
CMS proposes Medicare coverage for transcatheter tricuspid replacement
Edwards submitted a request for a national coverage determination in February, after receiving FDA approval for its Evoque device earlier that month.
By Elise Reuter • Updated Dec. 23, 2024 -
Robots, AI and PFA: The top medtech Q&As of 2024
From challenging market leaders to the growing influence of artificial intelligence, medical device executives had a lot to discuss this year. Check out MedTech Dive’s top Q&As of 2024.
By Ricky Zipp • Dec. 20, 2024 -
Heart valve developer Anteris launches IPO
IPOs in medtech have been sparse, but some analysts said improving market conditions could support a revival of offerings in 2025.
By Susan Kelly • Dec. 20, 2024 -
BD receives FDA warning letter over quality system violations
Inspectors found 111 open tickets for software defects categorized as catastrophic or severe patient harm. Several safety complaints were also listed that BD didn’t report within the required timeframe.
By Nick Paul Taylor • Dec. 20, 2024 -
Medical device recalls under the spotlight in 2024
Medtech regulators and watchdogs took a closer look at recalls in 2024. Here’s a recap of notable recalls and product safety actions in the past year.
By Elise Reuter • Dec. 19, 2024 -
Boston Scientific updates cryoablation catheter instructions after 4 death reports
The update followed a higher than expected number of reports of esophageal injury after catheter ablation procedures for atrial fibrillation.
By Nick Paul Taylor • Dec. 19, 2024 -

 Retrieved from Merit Medical on December 17, 2024
Retrieved from Merit Medical on December 17, 2024
Merit Medical president to resign after unspecified conduct allegations
Joseph Wright’s resignation is effective in January, and CEO and founder Fred Lampropoulos has been reappointed president.
By Susan Kelly • Dec. 18, 2024 -
Dexcom adds generative AI to over-the-counter CGMs
Dexcom this week began rolling out the feature, which analyzes data to provide personalized tips and recommendations.
By Elise Reuter • Dec. 18, 2024 -
Trump’s return to office brings uncertainty for medtech industry
With about one month before inauguration day, check out a roundup of MedTech Dive’s coverage on what President-elect Donald Trump’s new administration means for medical device companies.
By Ricky Zipp • Dec. 18, 2024 -
Medtech firms have cut thousands of jobs this year. Will layoffs continue in 2025?
Industry experts said profit pressures and M&A contributed to job cuts. They said layoffs will likely continue into next year, but some expect the cuts to slow.
By Elise Reuter • Dec. 18, 2024 -
Boston Scientific pacemaker recall tied to 832 injuries, 2 deaths
Certain Accolade pacemakers can permanently enter safety mode, which limits the devices’ ability to treat patients properly and requires early replacement.
By Ricky Zipp • Updated Feb. 25, 2025