Diagnostics: Page 4
-
J&J, Abbott and Intuitive start medtech earnings season
The companies’ financial results will offer a glimpse at the current health of medtech markets.
By Nick Paul Taylor • July 16, 2024 -
House committee tells FDA to suspend lab developed test rule
Lawmakers said the final rule carries “the risk of greatly altering the United States’ laboratory testing infrastructure.”
By Nick Paul Taylor • July 15, 2024 -
 Explore the Trendline➔
Explore the Trendline➔
 Sitthiphong via Getty Images
Sitthiphong via Getty Images Trendline
TrendlineMedical device industry continues to turn to AI
While the industry continues to embrace artificial intelligence, there are still questions about how the new technologies need to be regulated and if they are effective.
By MedTech Dive staff -
Deep Dive
FDA’s lab developed test rule could be first check on agency’s power post-Chevron
The Supreme Court’s decision to overturn the Chevron doctrine would make it easier to challenge agency regulations, such as the LDT final rule.
By Susan Kelly , Elise Reuter • July 11, 2024 -
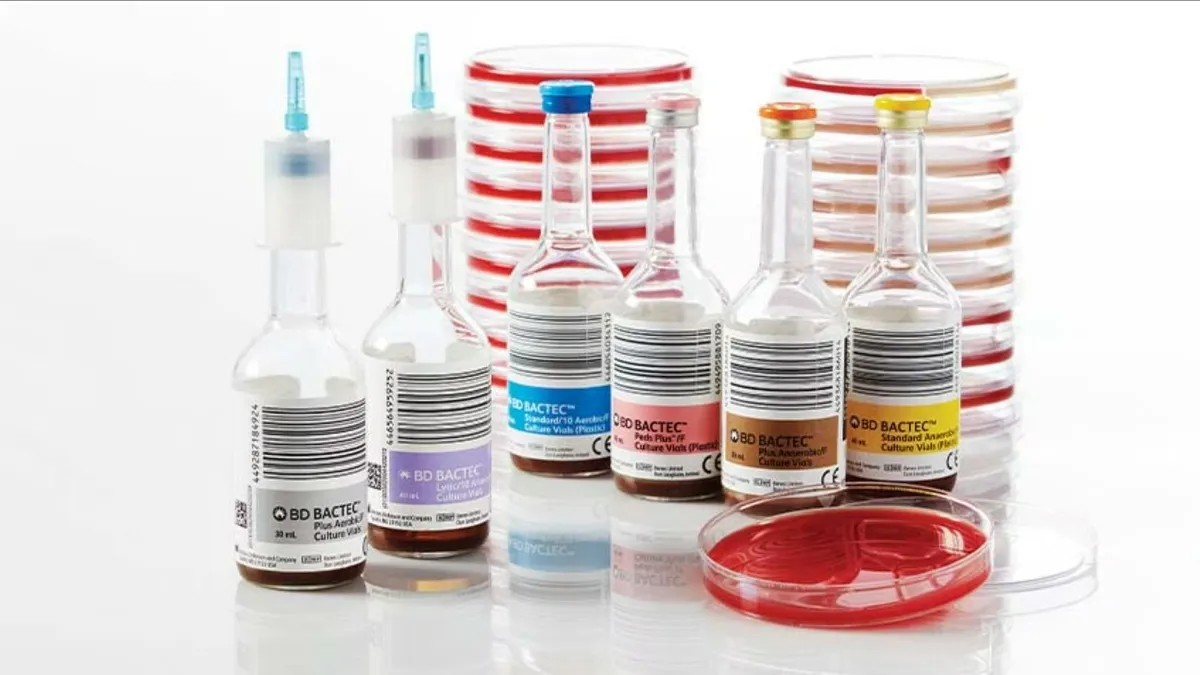
FDA warns BD blood culture bottle shortage could impact diagnosis
BD said reduced availability of blood culture media bottles due to supplier issues has prevented it from meeting global demand.
By Nick Paul Taylor • July 11, 2024 -
Quest to buy Lifelabs for $985M
Buying the Canadian laboratory company opens a new front in Quest’s rivalry with Labcorp and marks its third takeover in as many months.
By Nick Paul Taylor • July 8, 2024 -
Roche sues Foresight and Stanford, alleging use of trade secrets
Roche said Stanford applied for patents relating to cancer detection and monitoring technology using its confidential information.
By Nick Paul Taylor • July 3, 2024 -
Cepheid wins FDA nod for fingerstick hepatitis C test
Patients could be diagnosed and potentially receive treatment at the same healthcare visit.
By Nick Paul Taylor • June 28, 2024 -
FDA partners with Gates Foundation to support breath-based diagnostic development
Through a $1.9 million Gates Foundation grant, the collaborators will create methods to help developers make tests that can easily be deployed to rural and remote areas.
By Nick Paul Taylor • June 27, 2024 -
Illumina completes Grail spinoff
Nearly four years after Illumina announced a deal to buy Grail, the liquid biopsy maker will debut on Nasdaq Tuesday as a standalone company.
By Nick Paul Taylor • June 25, 2024 -

 Retrieved from Dermtech on June 20, 2024
Retrieved from Dermtech on June 20, 2024
Dermtech files for Chapter 11, lays off 20% of staff
The diagnostic test maker will continue to hunt for a buyer for its skin cancer detection patch.
By Nick Paul Taylor • June 20, 2024 -
Agilent to cut 3% of employees as sluggish lab equipment market persists
An Agilent spokesperson said the layoffs follow job cuts announced late last year amid a slower-than-expected market recovery.
By Susan Kelly • June 14, 2024 -
Danaher names Martin Stumpe as chief data and AI officer
Martin Stumpe will take over the new position after previously working as Tempus’ chief of AI and the head of Google’s pathology team.
By Nick Paul Taylor • June 6, 2024 -
Saliva test identifies men at high risk of prostate cancer
Data presented at the American Society of Clinical Oncology meeting suggest the test can support prostate cancer diagnosis in people who are missed by other screening methods.
By Nick Paul Taylor • June 5, 2024 -
Illumina board approves Grail spinoff
Leerink Partners analyst Puneet Souda said Grail, with a cash burn of $532 million in 2023, is likely to remain “a cautionary tale in diagnostics for years to come.”
By Susan Kelly • June 4, 2024 -
Illumina names Everett Cunningham as chief commercial officer
Cunningham will join Illumina from Exact Sciences to end the six-month search for a permanent leader.
By Nick Paul Taylor • June 4, 2024 -
Lab industry sues to stop FDA’s new LDT rule
The American Clinical Laboratory Association argues the agency does not have the authority to enforce stricter standards on lab developed tests.
By Susan Kelly • May 30, 2024 -
FDA panel backs Guardant’s blood test for colon cancer
“The favorable panel vote does not by any means imply this test will become an overnight success,” wrote William Blair analyst Andrew Brackmann.
By Susan Kelly • May 24, 2024 -
Cue Health to lay off remaining employees, shut down
Once a COVID-testing darling, Cue will close after raising hundreds of millions of dollars and striking high-profile partnerships with Google and the NBA during the pandemic.
By Elise Reuter • May 22, 2024 -
Guardant colon cancer test struggles to detect non-cancerous tumors, FDA says
Agency scientists highlighted the risk in materials posted ahead of a Thursday advisory panel meeting for the Guardant Health blood test.
By Susan Kelly • May 22, 2024 -
EMA updates advice on drug-device combinations under MDR, IVDR
The European Medicines Agency answered six new questions to clarify topics such as how companies can get advice on borderline products.
By Nick Paul Taylor • May 22, 2024 -
BD wins FDA approval for cervical cancer screening self-collection kit
Patients can self-collect the samples in locations such as retail pharmacies, which could encourage more people to get tested.
By Nick Paul Taylor • May 17, 2024 -
FDA advises against using Cue Health COVID-19 tests, sends warning letter
Agency inspectors found Cue made unauthorized changes to its COVID-19 kits that “reduced the reliability of the tests.”
By Nick Paul Taylor • May 14, 2024 -
Pacific Biosciences to lay off 195 employees as part of cost cuts
More than half of the job losses stem from the closure of the genome sequencer’s San Diego office amid an ongoing industry slump.
By Susan Kelly • May 14, 2024 -
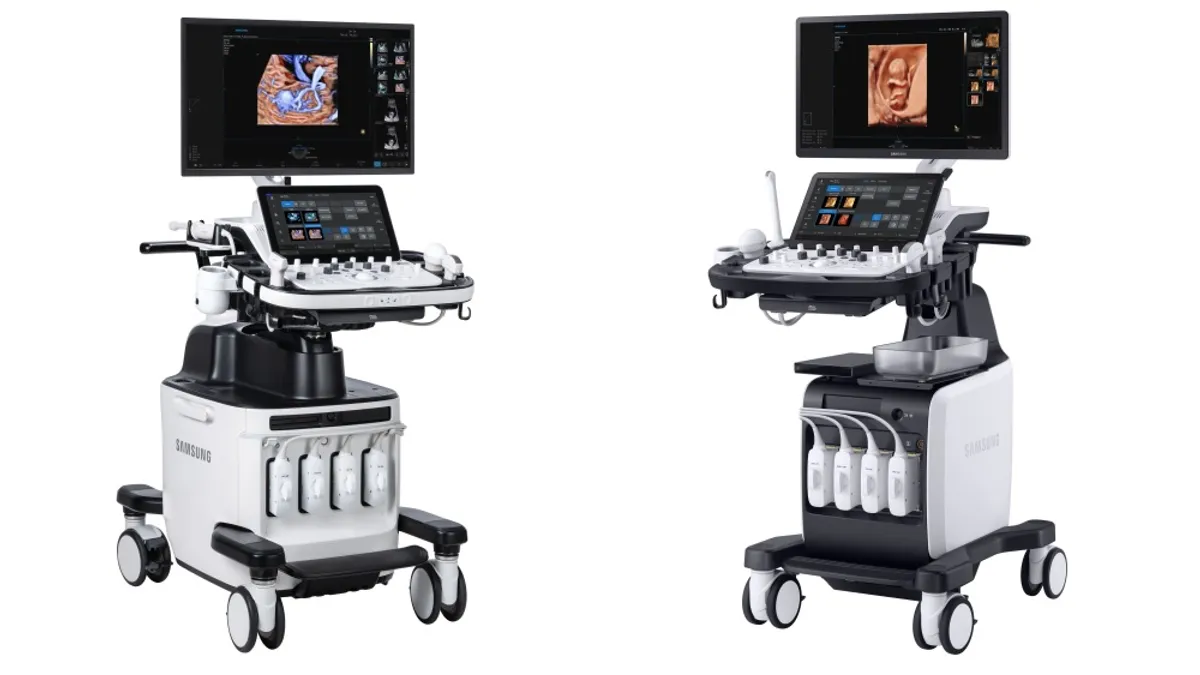
Samsung Medison to buy AI ultrasound company Sonio
Sonio’s software uses deep learning to improve image quality. Its software will remain compatible with all ultrasound machines, the company said.
By Elise Reuter • May 8, 2024 -
Geneoscopy wins FDA approval for Labcorp-partnered colon cancer test
Evercore and Cowen analysts expect Colosense to have a limited impact on Exact Sciences’ rival Cologuard test.
By Nick Paul Taylor • May 8, 2024