Clinical Trials: Page 10
-
Medtronic pits TAVR device against Edwards Sapien 3, eyes challenge to Abbott's MitraClip
The company appears to be doubling down on structural heart competition with a series of investments in the transcatheter aortic valve replacement and mitral valve repair markets.
By Nick Paul Taylor • Oct. 15, 2020 -
TCT 2020: Abbott devices, TAVR systems headline late breaker lineup
The five-day Transcatheter Cardiovascular Therapeutics event kicked off Wednesday.
By Susan Kelly • Oct. 12, 2020 -
Medtronic shares early data TAVR works in low-risk bicuspid patients
Authors of the short Medtronic-sponsored trial, published in JAMA Cardiology, suggest the valve may be free of problems that caused early studies of the device in bicuspid disease to report worse in-hospital outcomes.
By Nick Paul Taylor • Oct. 8, 2020 -
Deep Dive
5 ways COVID-19 is reshaping the medtech industry
Remote tech, clinical trials and marketing are a few of the areas disrupted by the pandemic this year — with changes that look set to stick around.
By Maria Rachal , Greg Slabodkin • Oct. 1, 2020 -
Abbott resurrects resorbable scaffold tech for below-the-knee trial
The medtech previously stopped selling the technology amid evidence it increased the risk of major adverse cardiac events, but recent data has revealed a new BTK opportunity.
By Nick Paul Taylor • Sept. 4, 2020 -
FDA proposes adding patient voice in device evaluation
The draft guidance comes nearly two years after an advisory committee prioritized greater inclusion of the patient perspective in device design, development and assessment. Two more public meetings are planned.
By Susan Kelly • Aug. 31, 2020 -
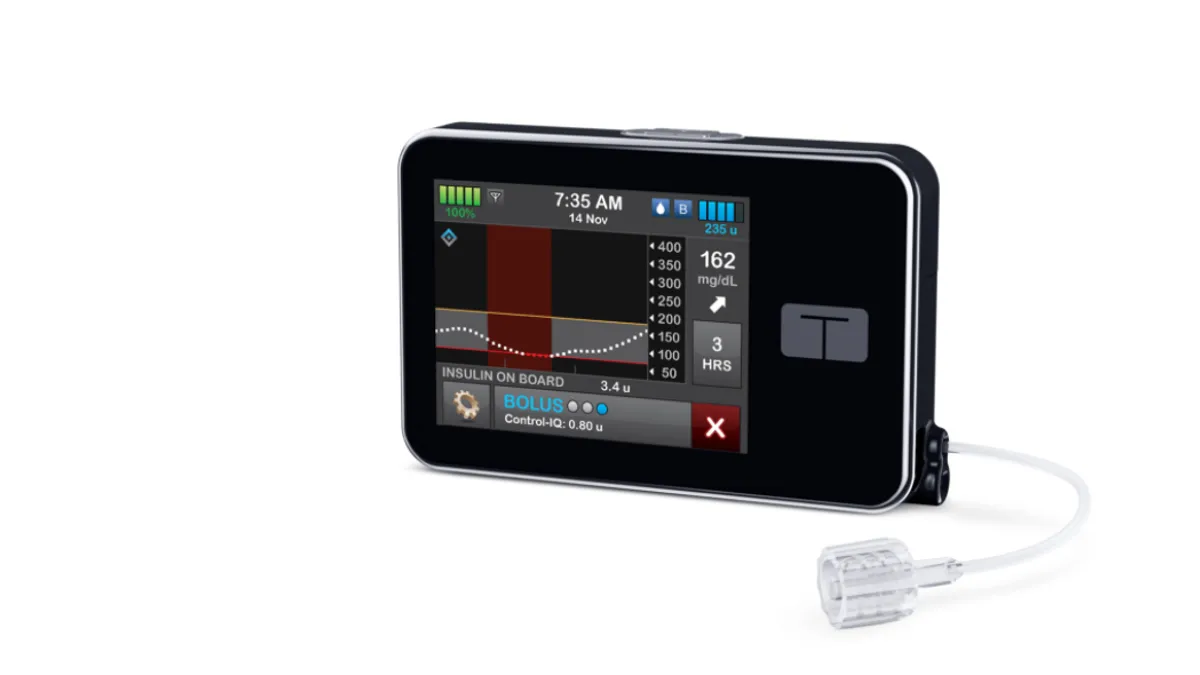
Tandem's artificial pancreas improves glucose control in pediatric trial
Children treated with the closed-loop insulin delivery system spent longer in the target glucose range than peers who just received Dexcom G6 sensors and insulin, the study published in the New England Journal of Medicine found.
By Nick Paul Taylor • Aug. 27, 2020 -
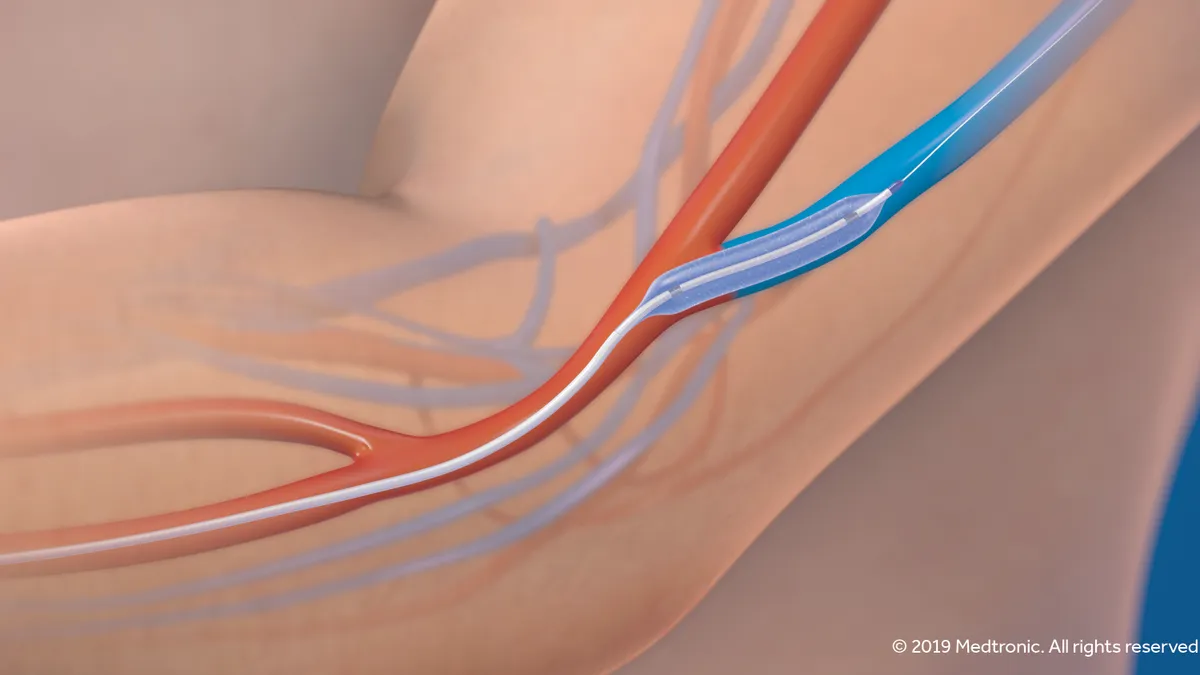
Medtronic hopes for renewed spotlight on dialysis access device with NEJM publication
The company-funded study in the New England Journal of Medicine comes amid widespread disruption of vascular access procedures, and nine months after Medtronic got FDA approval for its IN.PACT AV drug-coated balloon.
By Maria Rachal • Aug. 21, 2020 -
J&J drops planned 510(k) for Verb-Auris robot, targets 2022 clinical trial start
Talks with the FDA led Johnson & Johnson to opt against trying to bring its general surgery robotics platform to market via the 510(k) pathway. Launch delays buy market incumbent Intuitive some important time, analysts noted.
By Nick Paul Taylor • July 17, 2020 -
FDA proposes BPH device trial guidance changes as market heats up
The updates come as a swath of companies take aim at the benign prostatic hyperplasia opportunity, which medtechs like Boston Scientific and Teleflex have invested in more since FDA issued development guidance a decade ago.
By Nick Paul Taylor • July 14, 2020 -
Abbott lays out plan for US study of FreeStyle Libre 3
The sign of potential progress on the medtech's next version of its glucose monitoring technology comes about a month after Libre 2 gained FDA's green light, albeit without the OK to integrate with automated insulin delivery systems.
By Maria Rachal • July 13, 2020 -
Study design for CVRx device can serve as heart failure model: JACC editorial
Authors from Massachusetts General Hospital and Harvard Medical School say the adaptive design of the BeAT-HF trial used to support an FDA approval last year should be considered for future efforts on heart failure treatments.
By Susan Kelly • July 2, 2020 -
Medtronic renal denervation registry shows blood pressure improvements persist at 3 years
The update on Thursday from the medtech giant comes months after Symplicity clinical trial results boosted its long-running effort to win FDA approval.
By Nick Paul Taylor • June 26, 2020 -
Polares raises $40M to target competitive mitral regurgitation market
The medtech plans to finish pilot clinical studies of a transcatheter mitral valve hemi-replacement device, seeking to treat a slice of a population that's proven worthwhile for MitraClip maker Abbott.
By Nick Paul Taylor • June 26, 2020 -
Medtronic inks up to $337M in diabetes funding, Blackstone Life Sciences' 1st medtech investment
The announcement came in conjunction with the ADA's Scientific Sessions this weekend, which also featured data on its advanced hybrid closed loop system that will back an FDA premarket approval submission.
By Susan Kelly • June 15, 2020 -
Key data on Medtronic's 780G, Insulet's Horizon kick off ADA 2020
Abbott, Dexcom and Tandem will also present results at the virtual American Diabetes Association meeting that runs through Tuesday.
By Maria Rachal • June 12, 2020 -
Insulet's pivotal study back on track, setting up Horizon launch in early 2021
The diabetes device maker fixed a software glitch it found with the smartphone-controlled automated insulin delivery system three months ago. Abbott and Dexcom plan to make their CGM tech compatible with the system.
By Maria Rachal • June 5, 2020 -
JAMA reanalysis goes against 5-year Abbott stent study findings, upping odds of death
The JAMA Internal Medicine report raised doubts about the claim percutaneous coronary intervention is noninferior to bypass surgery and fueled a call for changes to guidelines.
By Nick Paul Taylor • June 3, 2020 -
Many heart device trials 'small and fragile,' come with misleading spin: JAMA study
A JAMA Internal Medicine review of 216 trials of invasive cardiovascular interventions also found commercial sponsor involvement linked to more distortion or misrepresentation of the data.
By Nick Paul Taylor • June 2, 2020 -
Investors bet diabetes tech immune to full brunt of COVID-19
Pure play diabetes device makers aren't clear yet on how economic uncertainty and the transition to telemedicine may affect new patient demand, but are outperforming the stock market by a long shot.
By Maria Rachal • May 19, 2020 -
Medtronic offers 10-year data, pivotal trial update on transcatheter pulmonary valves
A decade of data on the Melody valve found it more effective in adults than in pediatric patients. In a separate study, the in-development Harmony valve designed to treat severe pulmonary regurgitation met its primary endpoints.
By Susan Kelly • May 18, 2020 -
EC outlines clinical trial safety reporting between MDR, Eudamed start dates
Even with the recent delay to the date of application of the Medical Device Regulation, there will still be at least a 12-month period when the key Eudamed database is not fully available.
By Nick Paul Taylor • May 14, 2020 -
AtriCure treatment for persistent AFib shows superiority in study
The ablation procedure met study goals among patients with hard-to-treat forms of the heart rhythm disorder, which analysts say put it on track for FDA approval to address a market worth at least $1 billion.
By Susan Kelly • May 11, 2020 -
Thrive, chasing Grail with a cancer blood test, finds tumors in seemingly healthy women
A first-of-its-kind trial of 10,000 women supported the test's potential to catch some cancers early. But whether it can truly help people is not entirely clear.
By Ben Fidler • April 28, 2020 -
Novocure shares data from post-approval brain cancer device trial
As part of the virtual AACR 2020, researchers said data show use of a Novocure technology resulted in longer median overall survival than occurred with best standard of care chemotherapy.
By Nick Paul Taylor • April 28, 2020