Dive Brief:
- MiniMed, the diabetes tech firm spun out of Medtronic earlier this month, received Food and Drug Administration clearance for a smaller insulin pump.
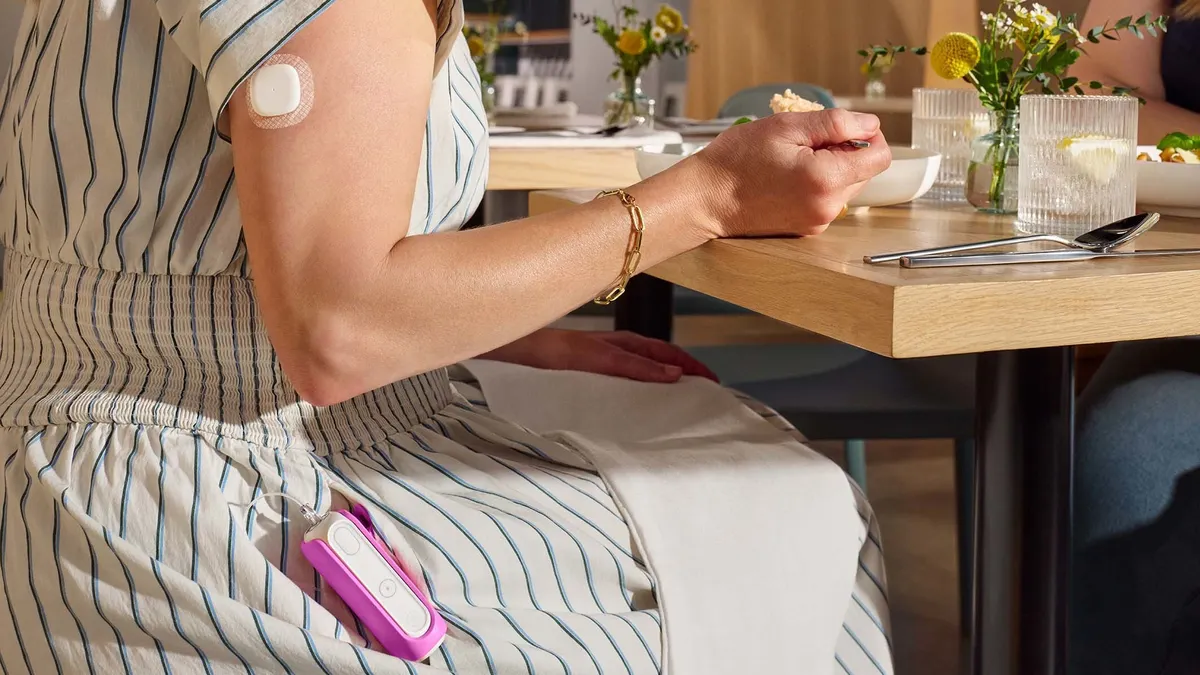
- The device, called MiniMed Flex, is about half the size of the company's previous 780G pump and is controlled using a smartphone.
- The new insulin pump is MiniMed’s first launch since the company went public in early March.
Dive Insight:
The smaller insulin pump is one of two showcased at the American Diabetes Association conference last summer. MiniMed plans to submit the other device, an insulin patch pump with a rechargeable battery, to the FDA this fall, Medtronic CFO Thierry Piéton said in a February earnings call.
MiniMed Flex has a 300-unit insulin reservoir and can pair with MiniMed’s latest continuous glucose monitors, including its Simplera Sync sensor and the Instinct Sensor made by Abbott. The device is indicated for people with Type 1 diabetes ages seven and up and Type 2 diabetes ages 18 and up.
MiniMed is one of the only diabetes tech firms to offer both insulin pumps and glucose sensors. The new company is banking on this approach in a competitive market.
“Securing FDA clearance for our first product as a standalone, public company — just one week after our IPO — marks a major milestone and underscores our commitment to delivering the breakthrough innovation our customers deserve,” CEO Que Dallara said in a statement.
MiniMed plans a limited launch of the new system this spring, with a broader commercial launch slated for this summer. Users of the company’s MiniMed 780G system can upgrade to MiniMed Flex for free at commercial launch.