Dive Brief:
- JenaValve has received premarket approval from the Food and Drug Administration for its transcatheter heart valve to treat symptomatic, severe aortic regurgitation in patients who are at high risk for surgical valve replacement.
- The Trilogy valve is now the first transcatheter device with a dedicated indication for the condition, the company said Wednesday.
- Edwards Lifesciences in January canceled its planned acquisition of JenaValve after the Federal Trade Commission challenged the $945 million deal, arguing it would combine the only two companies conducting U.S. clinical trials for transcatheter aortic valve replacement devices to treat aortic regurgitation.
Dive Insight:
Aortic regurgitation is a potentially fatal heart condition in which the valve does not close properly, allowing blood to leak backward into the heart. JenaValve said symptomatic, severe aortic regurgitation is significantly undertreated.
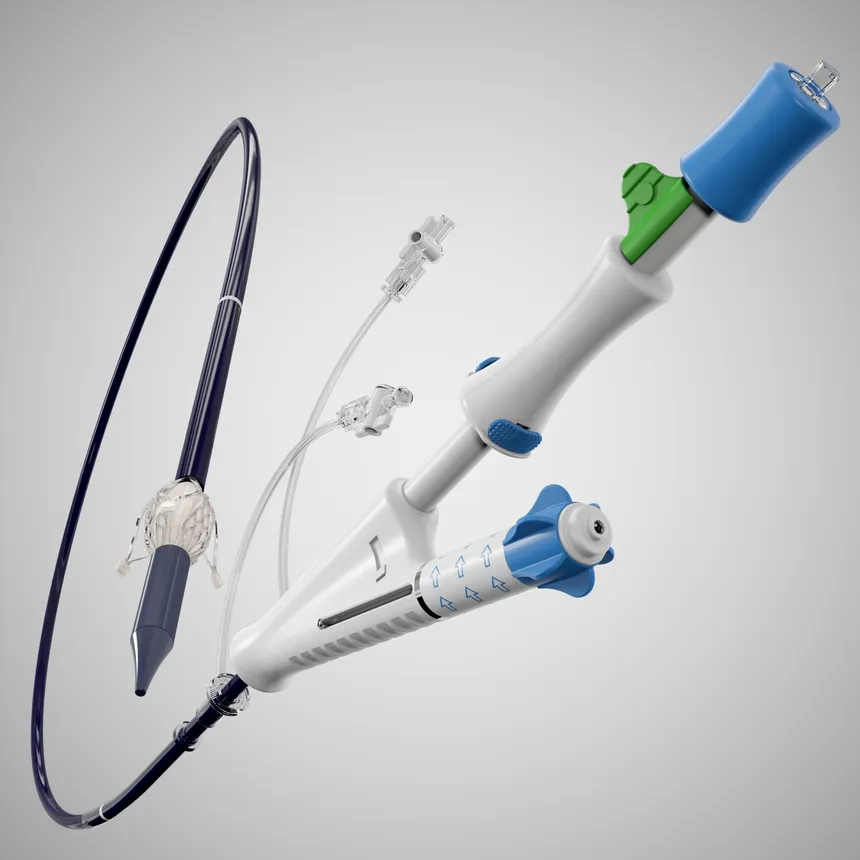
The approval was supported by results from the ALIGN-AR pivotal trial, which showed the Trilogy system is safe and effective for treating patients with the condition who are at high risk for surgery. The device has three locators that attach to the patient’s native aortic leaflets and are designed to enable precise positioning for implantation.
Trilogy has received Europe’s CE mark to treat both aortic regurgitation and stenosis, where the valve opening narrows and restricts blood flow.
Edwards, whose Sapien TAVR devices treat aortic stenosis, is also working to develop a regurgitation treatment. In 2024, Edwards acquired JC Medical, whose transcatheter valve is being studied in the JOURNEY pivotal trial.
Edwards ended its plans to acquire JenaValve after a U.S. district court granted the FTC’s motion for an injunction blocking the transaction.