Dive Brief:
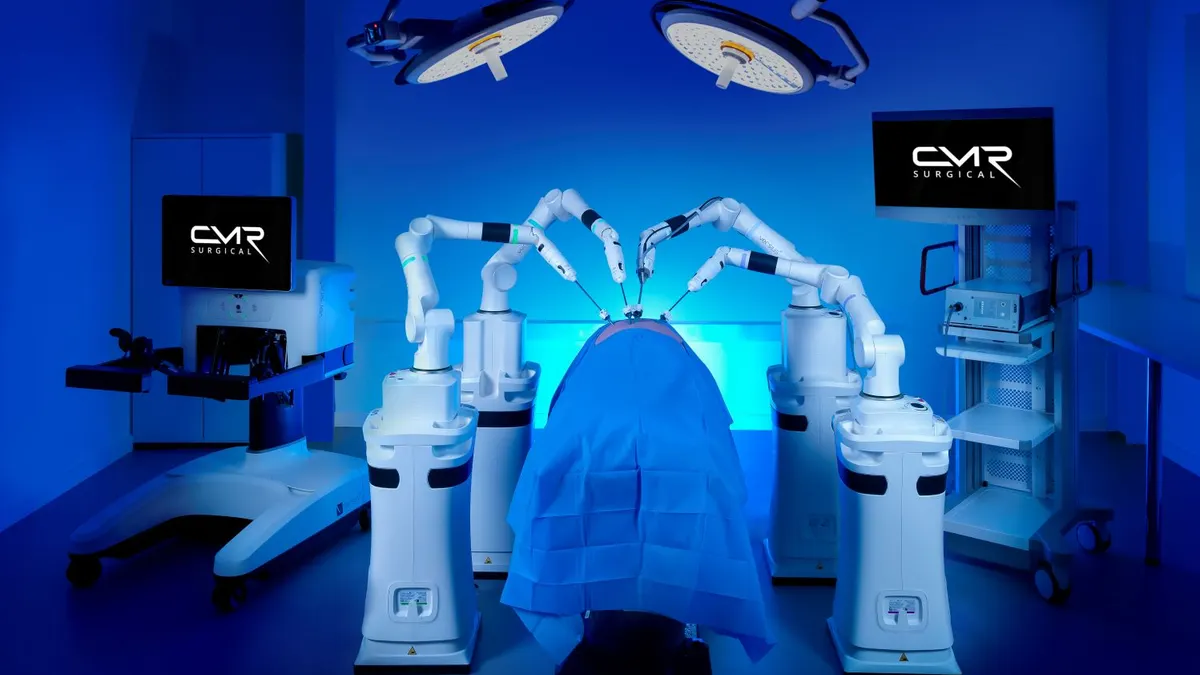
- CMR Surgical said Thursday that global adoption of its Versius surgical robot is accelerating across multiple clinical specialties as the company prepares to expand into the U.S. More than 45,000 patients worldwide have now been treated with a Versius robot, CMR said, up from the more than 40,000 reported in December.
- CMR obtained Food and Drug Administration clearance in December for Versius Plus, its next-generation system, for use in gallbladder removal surgery. More indications are planned.
- The Cambridge, England-based company provided the update in connection with its attendance at the annual meeting of the Society of American Gastrointestinal and Endoscopic Surgeons in Tampa, Florida, where it is formally introducing the robot to U.S. surgeons this week.
Dive Insight:
As more surgical robots are cleared in the U.S. — including Medtronic’s Hugo system, which also received the FDA’s nod in December — CMR claims to have the second most widely used platform in the world. Robotic surgery pioneer Intuitive Surgical remains at the top.
The growing momentum reflects surgeons’ confidence in the platform, CEO Massimiliano Colella said in a statement. The system is currently used in hospitals in more than 30 countries, in specialties that include general surgery, colorectal surgery, urology, gynecology and thoracic surgery.
“What’s notable here is the scale of experience behind Versius, with tens of thousands of procedures contributing to a growing body of clinical understanding,” Francesco Bianco, a physician in the University of Illinois Chicago’s division of general, minimally invasive and robotic surgery, said in a statement. “That level of real-world use, and the data that come with it, is important as surgeons evaluate new technologies entering the U.S. market.”
Colella said the U.S. debut of Versius marks a “watershed moment” for the company, which is beginning to form partnerships with hospitals and ambulatory surgery centers ahead of a broader rollout.